Since the coronavirus pandemic became a serious issue for all countries around the world, experts have been calling for a “contact-tracing app” that will able to track infected people via smartphones. Now tech giants like Google and Apple have come together with technology that might make this system a real-life possibility. All this- while preserving the privacy of the people using it.
Similarly, pharmaceutical giants like Sanofi and GSK announced that they have signed a letter of intent to create an adjuvanted vaccine to fight the coronavirus using innovative technology and science from both companies in order to address the current pandemic.
All in all- tech companies will help contain the spread of the virus, while pharmaceutical corporations will help cure those affected. While excellent in theory, how will these technologies work? Let’s elaborate.
How does contact-tracing work?
Both Apple and Google have announced a project to create the groundwork for a blue-tooth based contact tracing apps- it will run on both iOS and Android phones. They will release an API around next month that apps from all public health organizations can connect to. The API will let those apps use the phones’ Bluetooth radios (which have 30 feet range) – to keep a track of whether the smartphone use has come into contact with someone who is infected with the coronavirus. Once they are alerted- they can self-isolate or contact healthcare professionals.
Most importantly, Apple and Google will not track user locations or even collect any personally identifiable data that could be stored on a server. In this system, the operating system level Bluetooth tracking will permit the users to opt-in to a Bluetooth-based proximity detection scheme. Their smartphone will alert them with Bluetooth signals to others in the close distance while also listening for signals from nearby smartphones.
If two phones spend a few minutes within a range of each other- they record contact with other phones – exchanging one of a kind rotating identifier ‘beacon’ numbers that are established on the basis of the keys stored on each device. Thus public health app developers can ‘tune’ both the closeness as well as the time necessary to qualify contact- based on the current data about how COVID-19 spreads.
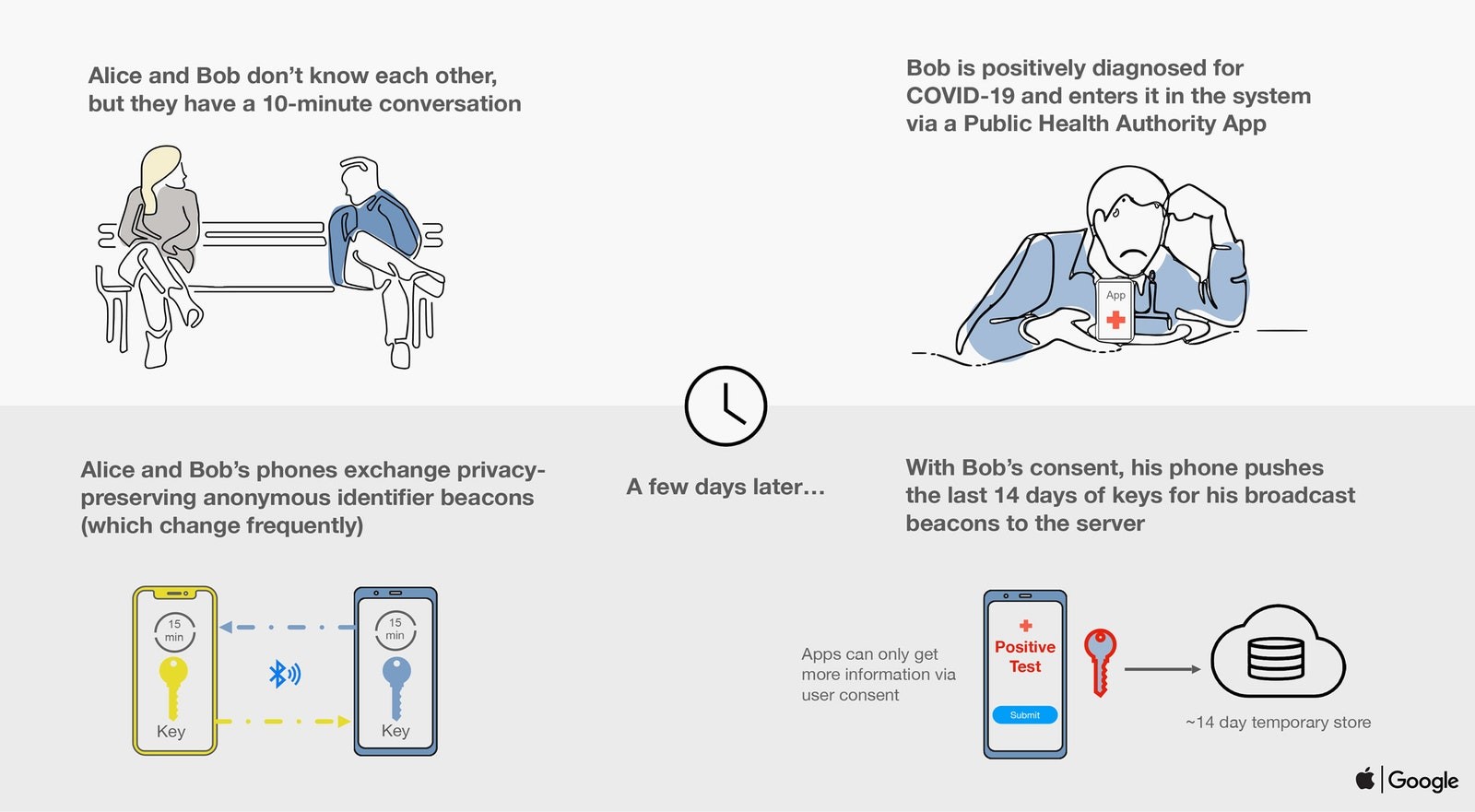
If a user later becomes diagnosed with COVID 19- they will alert the app by selecting an option. The application will then upload the last two weeks of keys to the server – this will create their recent ‘beacon’ numbers and send them to other phones in the system. Therefore, if someone else’s phone finds the same beacon numbers stored in their phone (matching to those sent) – they would be alerted that they have been in contact with a potentially infected person. With this information spread of the virus can be contained.
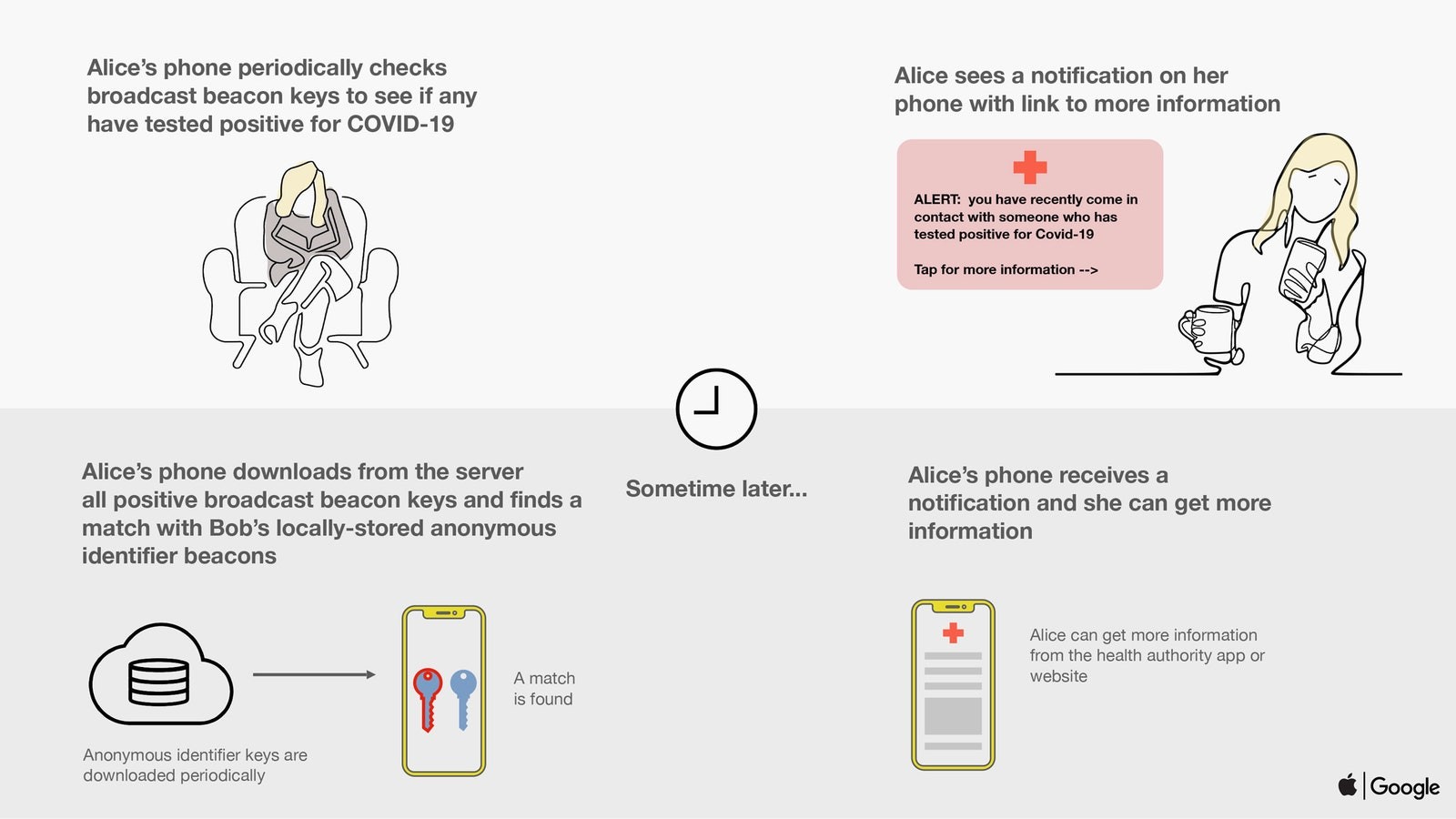
Of course, this is system has its limitations in itself like for e.g. infected person leaving traces on virus on surfaces which cannot be tracked by cellphone contacts or people’s willingness to share information regarding their diagnosis. Contact tracing, however, is the first step in the right direction.
GSK and Sanofi join hands for vaccine collaboration in the fight against COVID-19
Sanofi and GSK- two of the most major pharmaceutical companies have also decided to collaborate to combine innovative technologies to develop an adjuvanted COVID-19 vaccine. The companies announced that Sanofi will provide its S-protein COVID-19 antigen based on recombinant DNA technology. This technology has come up with an “exact genetic match to proteins found on the surface of the virus, and the DNA sequence encoding this antigen has been combined into the DNA of the baculovirus expression platform, the basis of Sanofi’s licensed recombinant influenza product in the US”.
Meanwhile, GSK will provide it’s tested and proved pandemic adjuvant technology. Using adjuvant can successfully prove to be advantageous in a pandemic as it may decrease the amount of vaccine protein needed per dose- permitting more vaccine doses to be produced and thereby, contributing to protecting an increased number of people.
A combination of adjuvant and protein-based antigen is well established and utilized in many vaccines. An adjuvant is added in some vaccines to advance the immune response and has proved to create a longer and stronger immunity against diverse infections. It can also enhance the possibility of delivering an effective vaccine that can be manufactured at a scale.
“Strategic alliances among vaccine industry leaders are essential to make a coronavirus vaccine available as soon as possible. Development of the adjuvanted recombinant-based COVID-19 vaccine candidate holds the potential to lower the vaccine dose to provide vaccine to a greater number of people to end this pandemic, and help the world become better prepared or even prevent future coronavirus outbreaks” says BARDA Director, Rick A. Bright, Ph.D.












